Augsburg University has a strong commitment to training undergraduates in research. The University funds or administers several research-intensive training programs both in the summer and during the academic year. Students majoring in Biology work with one of several research mentors in the department as well as with mentors in Physics, Chemistry, Mathematics, and Psychology. Students are encouraged to complete their research training early in their undergraduate careers so as to whenever possible remain engaged for a sustained period either on campus or at research universities.
For more information about on campus support and resources, visit the Undergraduate Research and Graduate Opportunity site.
Ongoing faculty research projects are listed below.
Jennifer Bankers-Fulbright
 Dr. Bankers-Fulbright’s research in the Airway Inflammation Research (AIR) Lab focuses on the interactions of human airway cells with pathogens and immune cells.
Dr. Bankers-Fulbright’s research in the Airway Inflammation Research (AIR) Lab focuses on the interactions of human airway cells with pathogens and immune cells.
Currently, Dr. B is focusing on testing the effects of human airway lung secretions on Pseudomonas aeruginosa. P. aeruginosa is a common bacteria that often contaminates hospitals but usually doesn’t cause serious disease. However, the lungs of patients with the genetic disorder, Cystic Fibrosis (CF), are extremely susceptible to P. aeruginosa colonization and often die from these lung infections.
Dr. B’s hypothesis is that secretions from normal airway cells inhibit this colonization by decreasing P. aeruginosa survival, motility, and biofilm formation. Conversely, she predicts that secretions from CF airway cells are altered in such a way that P. aeruginosa survival, motility and biofilm formation are unaffected, which allows for colonization of CF lungs. To test this hypothesis, AIR Lab students are examining the effects of harvested secretions from normal and CF airway cells on P. aerugionsa growth, flagellar motility, and biofilm formation in vitro.
Ideal for Sophomores and Juniors who are interested in graduate school and thinking about pursuing an M.S. or Ph.D. in the Biomedical sciences. (Research students must have completed Bio253 before working in the AIR Lab)
- Basic Lab Techniques
- General lab safety and accurate lab notebook use
- General Lab Techniques (pipetting, making solutions, pH)
- Mammalian cell tissue culture w/light microscopy
- Microbiological culture and assays
- Data analysis and graphing
Matthew Beckman
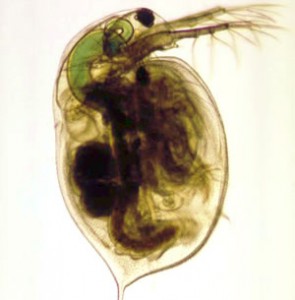
Our work is split between two very different projects. First, we want to know how some organisms have evolved to have only one eye (cyclopia). To address this question, we study the developmental genetics of cyclopia in Daphnia magna and related branchiopods by using Molecular Biology and high-resolution microscopic imaging methods. Second, we study the effects of manganese on cellular function. This work makes use of Daphnia magna to assess the motor behavioral effects of manganese and aims to find small molecules that may prevent or treat the detrimental effect of this metal. Also, we use human neuroblastoma cells to study the mitochondrial damage elicited by manganese. To do this work we use behavioral assays in Daphnia and high-resolution fluorescence imaging of cultured neuroblastoma cells with a specific focus on mitochondrial structure and dynamics.
Bill Capman
My research deals with developing and refining captive breeding and larval rearing methodologies for marine (saltwater) fish and invertebrates. Until fairly recently, the majority of coral reef fish and invertebrates were considered to be impossible to breed in captivity, with the major challenges being meeting the unique physical and dietary needs of planktonic larval stages adapted for life drifting in the open ocean. In the past, nearly all marine life sold for aquariums was collected in the wild, sometimes with undesirable environmental consequences. In recent decades though great past advances have been made in this field, with tremendous breakthroughs in breeding coral reef fish occurring in just the last six years or so. Over the past few years students in my lab have worked with me in the breeding of a unique species of clownfish, Yellow Belly Blue Damselfish (whose tiny larvae are very challenging to rear), two species of Peppermint Shrimp, and most notably Red Spine Blue Tuxedo Sea Urchins. in addition, we have multiple other species that are spawning in our coral reef aquariums that we would like to work with (including coral beauty and Lamark’s angelfish). Our research with rearing urchin larvae in particular has involved investigating the effects of diet on larval development, and we have also greatly refined larval rearing methodology. We now have produced three successive generations of captive bred urchins, and we have become one of the main breeders of these urchins in the U.S. (if not the world).
David Crowe

Along with collaborators at the University of Minnesota, I have been studying neural and genetic deficits underlying schizophrenia. Schizophrenia is a debilitating disorder affecting millions of people. Current pharmaceuticals are somewhat effective in treating the positive symptoms (e.g. hallucinations, psychosis), but are ineffective in treating cognitive deficits (e.g. memory, attention, cognitive control problems). Improving treatment of schizophrenia likely requires a deeper understanding of the patterns of neural activity manifest in the disorder. My collaborators and I study the electrical activity of neurons in two animal models of schizophrenia. In a pharmacological model, we block NMDA receptors with drugs known to produce symptoms associated with schizophrenia. In a genetic model, we study mice who are missing genes that are associated with the disorder. Students working with me analyze data from these experiments using computer programming.
Emily Schilling
Freshwater ecosystems are among the most threatened on Earth. My research addresses a pressing need to understand how these ecosystems function in natural and human-altered landscapes. Much of my work has specifically focused on understanding both natural and anthropogenic factors that influence freshwater species’ distributions and community structure. To do this, I have often used geospatial analytical tools like GIS for exploring biogeographic patterns in freshwater species’ distributions and answering questions related to freshwater species movement. I examine ways that humans interrupt biogeographic barriers to species movement, for example by stocking non-native gamefish to provide wilderness fishing opportunities, and how this affects native freshwater communities. This work has application beyond the wilderness, to places closer to home in our local urban lakes and streams. These places that might be familiar to the public have historically been under-represented in ecology, and their role in environmental resiliency is gaining attention.
Jason Tan
Phages, which are viruses that infect bacteria, can be used as a tool to alter the composition and metabolic capabilities of microbial communities that reside in our bodies and in the environment. For example, bacteria in nature may be “genetically reprogrammed” by introducing phages as agents of horizontal gene transfer that naturally spread, leading bacteria to better metabolize and detoxify harmful compounds (i.e. bioremediation); alternatively, phages or phage proteins can be purified and given to patients (human or animal) to eliminate antibiotic-resistant bacterial infections (i.e. phage therapy). In our lab, students gain valuable experience in solving complex biological problems by applying a combination of genetic, biochemical, and computational approaches to (1) study molecular mechanisms underlying regulation of gene expression during different steps in the phage life-cycle, or (2) identify and characterize novel phages that are present in bacterial communities that shape various ecosystems in Minnesota. Join us in our “phage safaris” as we seek to better understand the many ways in which phages shape biological processes within our own bodies, as well as in external ecosystems that we may be part of on planet Earth (and beyond)!
Leon van Eck
My lab uses the cereal crop barley as a model to study biotic interactions with phloem-feeding insects called aphids. Since plants cannot run away when under attack, they must stand and fight using Biochemistry. Through millions of years of co-Evolution, plants have developed highly sophisticated molecular surveillance and genetic response machinery to detect and combat pests and pathogens. However, the molecular arms races that constitute plant-insect and plant-microbe interactions are still poorly understood. If we wish to sustainably produce food in a future where climate change and outbreaks of pests and diseases are a major concern, without relying on expensive and hazardous agrochemicals, then we must turn to plant genetics for crop improvement. I am particularly interested in crop wild relatives–the undomesticated cousins and ancestors of our food crops–as a source of potentially useful genes. Through my collaborators at the UMN Stakman–Borlaug Center for Sustainable Plant Health, I have access to the Wild Barley Germplasm Collection, which is extensively used in student research. The Molecular Biology projects students do in my lab include quantitative expression analysis of plant stress-related genes, insect feeding electrophysiology and fecundity analysis, histochemical staining and microscopy of plant tissues, bioinformatic mining of the barley genome, and microbiological analyses of the phytobiome.
